 Since the given mass is less than half of the molar mass of chlorine, the resulting number of molecules is less than half of Avogadro's number. The lessons learned in this post will continue through the study of chemical reactions and equations.\) We also learned how to calculate the molar mass of a compound using the periodic table and how to convert mass into moles. The given mass of K (4.7 g) is a bit more than one-tenth the molar mass (39.10 g), so a reasonable ballpark estimate of the number of moles would be slightly greater than 0.1 mol.  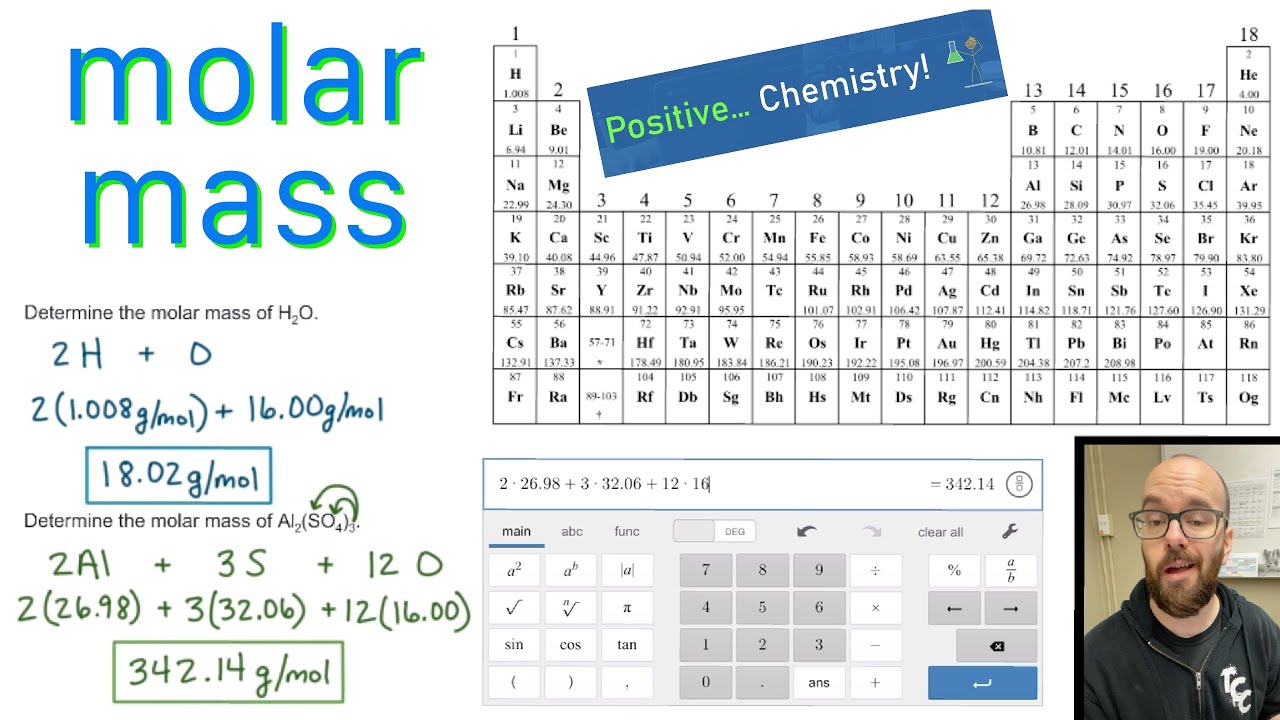
We learned about Avogadro’s number and how it relates to the mole, which is a unit used to express the amount of a substance. Formula: CaCl 2 Hill system formula: Ca 1 Cl 2 CAS registry number: 1 Formula weight: 110. Referring to the periodic table, the atomic mass of K is 39.10 amu, and so its molar mass is 39.10 g/mol. In conclusion, understanding molar mass is an essential concept in chemistry as it allows us to relate the amount of a substance to the number of particles present in it. Specifically, the number is defined as 6.022 \times 10^. There are 4 easy steps to find the molar mass of CaCl based on its chemical formula. The periodic table lists the atomic mass of carbon as 12.011 amu. The molar mass of any substance is its atomic mass, molecular mass, or formula mass in grams per mole. Example 3: Finding the Mass of a Number of MoleculesĪvogadro’s number is a fundamental constant that represents the number of particles (atoms, molecules, ions) in one mole of a substance. In each case, the number of grams in 1 mol is the same as the number of atomic mass units that describe the atomic mass, the molecular mass, or the formula mass, respectively.How to Find Molar Mass Using the Periodic Table Find: g CaCl 2: List other known quantities (molar mass is calculated by adding the atomic weight of each atom in the molecule) 1 ml CaCl 2 110.The Mole and its Relationship to Avogadro’s Number. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
